The following investigative report was contributed by @LisaMacRichards (a pseudonym). @LisaMacRichards works at a Canadian hospital and holds a Master of Science degree from the University of British Columbia. We’re very thankful for the diligent work she invested to research and write this article and we appreciate the opportunity to publish it here.
by @LisaMacRichards
Many people are surprised to find out that children with gender dysphoria are being prescribed puberty blockers as early as age ten, cross-sex hormones as early as age 12 or 13, and double mastectomies for girls as early as age 13 in countries throughout the world, including Canada. Some professionals and transgender advocates say that gender-dysphoric teens should be able to have genital surgeries and hysterectomies before age 18 on the basis of informed consent, with no mental health assessment required.
In Canada, transition-related care, including sex reassignment surgery is typically provided by specialized gender clinics. Provincial health insurers such as OHIP in Ontario and the PHSA in British Columbia require that providers align with the World Professional Association of Transgender Health Standards of Care (WPATH SOC). WPATH published its seventh version of the SOC in 2011, and is currently working on its eight version.
The amended OHIP criteria… align with the internationally-accepted standards of care for gender dysphoria, which are established by the World Professional Association of Transgender Health (WPATH).
Ontario Ministry of Health
The Canadian Mental Health Association references WPATH in its public website statement that medical transition for gender-diverse youth is the best way to treat the distress that arises when an individual’s body does not match their gender identity.
Whenever anyone questions the early social transition of children and adolescents with gender dysphoria or the use of medical interventions to align one’s body with their gender identity, the usual response from transgender advocates is that there is an international consensus of transgender treatment among professionals. In fact, the National Post recently published an article with a quote from a transgender activist saying “transition-related care is safe, effective, and supported by the entire mainstream of the medical community.”
Medical Guidelines
Guidelines are becoming increasingly important for busy medical practitioners who cannot stay abreast of all developments in every medical field. To address this, professional medical bodies and societies started to publish various guidelines in the 1990s, in order to clearly formulate best medical practices, to assure patients they are receiving appropriate care, and to standardize care so that most patients will receive the same care across many settings. However, many people started questioning whether medical guidelines are being developed using quality medical evidence and whether conflicts of interest could be influencing the recommendations.
Challenges in guideline development include limitations in the scientific evidence on which clinical practice guidelines (CPGs) are based, lack of transparency of development groups’ methodologies, questions about how to reconcile conflicting guidelines, and conflict of interests among guideline development group members and funders.
To address these problems, the Institute of Medicine (now called the National Academy of Medicine) wrote “Clinical Practice Guidelines We Can Trust”, published in 2011, which outlined trustworthy guideline development that minimizes bias and conflict of interest. Clinical guidelines in the US were kept in a central database at the National Guideline Clearinghouse (NGC), which was funded by the US Department of Health and Human Services. Having a government-funded, non-profit third party to analyze guidelines is crucial in order to assess the quality of clinical practice guidelines. However, in 2018, the Trump administration cut this funding, and the NGC has unfortunately been shut down. A non-profit patient safety organization, ECRI Guidelines Trust has stepped in.
Canada does not have its own third-party guideline clearinghouse. The Canadian Medical Association keeps a database of Canadian clinical guidelines, through a subsidiary called “Joule”. Guidelines International Network (GIN) also maintains a guideline library.
WPATH Guidelines Do Not Qualify for ECRI Trust Scorecard
ECRI provides a “Trust Scorecard” that rates the quality of the guidelines, based on evidence strength and the measures taken to reduce bias in the recommendations. After searching the ECRI database for transgender care guidelines, the only guideline posted was the “Endocrine Society Clinical Practice Guidelines of Gender-Dysphoric/Gender-Incongruent Persons, 2017”. It was not given a Trust Scorecard rating. The WPATH SOC was not included in the database.
When ECRI was contacted by this author and asked why WPATH guidelines were not included, and why the Endocrine Society guidelines did not have a scorecard rating, they responded in an email, saying that the reason the Endocrine Society guidelines did not meet inclusion criteria to be rated was because “Only a few of their recommendations were supported by the systematic review; the majority were not.” The reason WPATH was not included, ECRI stated, was because the guidelines were over five years old, and “did not use a systematic review to process”.
They did not use a systematic review to process.
ECRI
A search of the Canadian database yielded no results for transgender treatment. When contacted, Joule’s response was “In order to be included in our database, all guidelines have to be either developed or endorsed by an authoritative medical/healthcare organization. Neither the WPATH nor the Endocrine Society guidelines on transgender care have been endorsed by any Canadian organization(s) and therefore are not included in our database”. A search of the GIN library also yielded no results for transgender care.
Neither the WPATH nor the Endocrine Society guidelines on transgender care have been endorsed by any Canadian organization(s) and therefore are not included in our database.
Joule
Guidelines over five years old are at high risk of becoming clinically irrelevant due to new research findings, or changes in patient populations. Given the rapidly changing epidemiology and treatments in transgender care, seven-year-old guidelines are severely outdated in this field. One case in point is that when the WPATH guidelines were published in 2011, natal males were the primary group presenting to gender clinics in adolescence, but now males have been eclipsed by natal females 3 to 1.
Research on male adolescent clients cannot be generalized to females, an age-old fallacy in medicine. This makes the entire recommendation section for adolescent transgender care in the WPATH SOC dubious at best, irrelevant at worst. Investigation must be undertaken to determine why a new cohort has developed, and whether this group can be included in guidelines that were developed for a group with different histories and possibly different etiologies.
Conflict of Interest in Guideline Development and Committee Members
A systematic review of clinical guideline development in 2011 revealed 56 to 87% of the authors had a conflict of interest (COI) in all clinical practice guidelines. In “Guidelines We Can Trust”, the Institute of Medicine (IOM) states that a key factor in eliminating bias and conflict of interest (COI) is taking care in the composition of guideline committee members. Obvious financial COIs such as affiliations with pharmaceutical companies are a given.
There are other significant sources of conflict of interests, however, that also must be considered. According to IOM, examples of non-commerical financial activities include research grants and support from foundations or other nonprofit organizations for the person’s work or their institution. A person whose work or professional group is fundamentally jeopardized or enhanced by a guideline recommendation is said to have intellectual COI. Other intellectual COIs include authoring a publication or acting as an investigator on a peer-reviewed grant directly related to the recommendations under consideration.
The chair of a guideline development committee and most of the members should not have any conflict of interest, including income from clinical work. Often, however, in any given field, it is hard to find any experts who are not receiving income in their practices. The IOM guidelines allow for some members to have COIs, as long as they do not have voting privileges or make up over half the group. Ideally, the majority of guideline developers should be unconflicted methodologists, such as epidemiologists, statisticians, healthcare researchers, and/or “guidelineologists”, who lead the formulation of recommendations in collaboration with clinical experts.
Conflict of Interest in WPATH SOC Committee Members
When looking at the WPATH committee who worked on the current SOC document, a cursory examination of the members reveals that every one of the members have significant COIs. All of them either receive income based on recommendations in the guidelines, work at clinics or universities who receive funds from advocacy groups, foundations, or pharmaceutical companies who heavily favour a certain treatment paradigm, or have received grants and published papers or research in transgender care. The majority of the members are from the US, and six of them have affiliations with the same university–the University of Minnesota Program in Sexuality, which is primarily funded by a transgender advocacy organization (Tawani Foundation).
Eli Coleman, the committee chair for the WPATH SOC, who IOM guidelines stipulate should be completely free of conflict of interest, has his very position at the University of Minnesota funded by Jennifer Pritzer, a trans person and head of Tawani. In fact, there are press releases of Eli Coleman in 2017 thanking Jennifer Pritzer profusely for a generous donation, which adds up to 6.5 million dollars that Tawani has given to the university. Tawani also funded WPATH SOC development. Another advocacy group, Gender Identity Research and Education Society (GIRES) funded the translation of the SOC into various languages.

There was also an “anonymous donor” who supported guideline development. Did the anonymous funder want a certain outcome from the recommendations? Who knows?
Funding sources must be named, according to the IOM guidelines.
Three of the same committee members for the WPATH Guidelines, also served on the Endocrine Society guideline committee, which means intellectual COI is at play. Any recommendations based on faulty conclusions in the WPATH guidelines would potentially have been duplicated in the Endocrine Society guidelines.
Although pharmaceutical companies were not directly involved in WPATH SOC, they often donate to advocacy organizations, which could directly influence recommendations. Pharmaceutical companies often make large donations to universities as well.
At least four committee members are or were editors of transgender research journals. In the US, 50% of editors of research journals take industry payments, and yet they do not need to declare conflicts of interest. Editors have control over which articles are published, which can shape systematic reviews in the future. It is well established now that journal reviewers and editors are less enthusiastic to publish a study with unremarkable or negative results The WPATH SOC chair, Eli Coleman, is also an editor of a transgender journal and started another journal in the past.
WPATH has a history of being funded and pressured by transgender activists and transgender organizations. For example, during the 2018 WPATH conference when the version 8 SOC development was being discussed, Dr. Marci Bowers, a prominent transgender surgeon who performs genital surgery, stood up in the meeting and urged the SOC committee members to be “bold” in their recommendations. Many transgender people in the audience stood up and clapped.
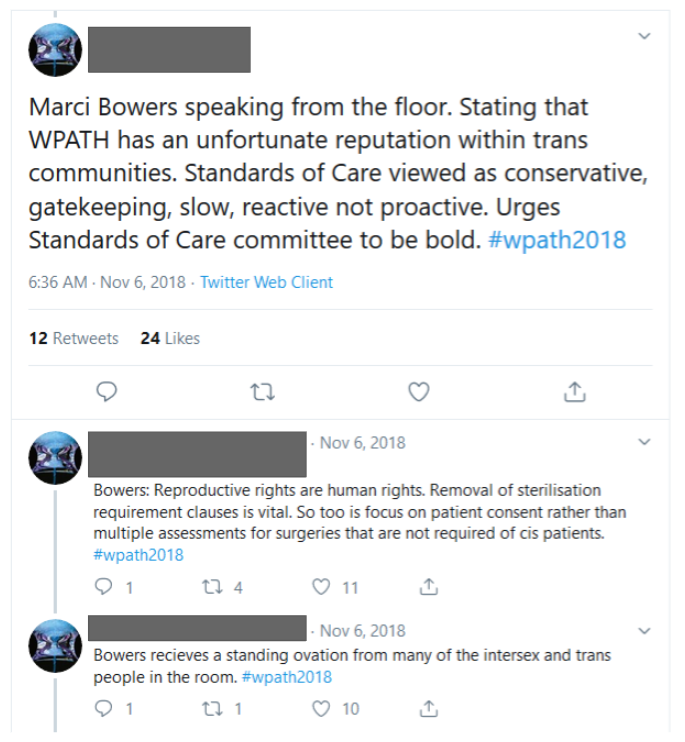
This surgeon is now on the WPATH Board of Directors.
At the 2017 USPATH/WPATH conference, activists protested and shut down a session with Dr. Kenneth Zucker, who endorsed a cautious approach to treating children with gender dysphoria. Rather than engaging with different viewpoints, WPATH agreed to cancel Dr. Zucker (who ironically was on the WPATH SOC committee) and apologize on their website for inviting him. WPATH is allowed to discuss only topics and engage with presenters that are approved by activists, which is the antithesis to a professional organization, or evidence-based inquiry.
WPATH is not the typical professional organization that develops clinical practice guidelines. WPATH is a hybrid professional and activist organization, where activists have become voting members and have served as president. In fact, it can be argued that WPATH is activist-led rather than evidence-led, as witnessed at their conferences. Their guidelines are just following what is being performed in gender clinics based on informed consent.
Lack of Systematic Review and Evidence for WPATH SOC
WPATH did not perform a systematic review to draw conclusions for their recommendations in the SOC. Perhaps WPATH already knew that there would be no evidence for their recommendations, as they state this numerous times throughout the guidelines. There are assertions such as:
To date, no controlled clinical trials of any feminizing/masculinizing hormone regimen have been conducted to evaluate safety or efficacy in producing physical transition.
WPATH SOC Version 7
They then proceed to recommend hormones with no rationale given.
The document is full of contradicting statements. For example, WPATH says:
“Gender dysphoria during childhood does not inevitably continue into adulthood. Rather, in follow-up studies of prepubertal children (mainly boys) who were referred to clinics for assessment of gender dysphoria, the dysphoria persisted into adulthood for only 6 to 23% of children.”
And then they later state: “Early use of puberty-suppressing hormones may avert negative social and emotional consequences of gender dysphoria more effectively than their later use would.”
So even though only 6 to 23% of gender dysphoric children will persist into adulthood, WPATH, with no rationale given, endorses suppressing puberty as soon as it starts.
In fact, no rationale is given as to why a medical model of affirmation is recommended in the first place.
They briefly state that conversion therapy is not successful. Conversion therapy is not defined but seems to be anything other than the medical model to treat gender dysphoria. The references they provide are three chapter books or case studies from the 1960’s that are no longer in print, and an article written in Dutch from 1984. Two of the titles refer to “aversion therapy”, which most people would agree is not ethical. However, counseling, cognitive behaviour therapy, or psychotherapy is not considered.
A literature search performed by ECRI shows that no systematic research or review has been conducted to see if any non-medical therapy could be successful, in order to avoid a lifetime of medication and surgeries. The assumption that thoughts and feelings are unchangeable, while the body should be changed, is the paradigm that is assumed in this document with no rationale given. A lifetime of off-label medications and multiple surgeries are assumed to be preferable to the option of considering treatment to help people be at peace in their natural bodies.
In light of the COIs described above, we can see that there are many benefits to a medicalized model for the committee members, both financially and for their careers. If evidence showed that a non-medical treatment for gender dysphoria was more effective, would committee members be inclined to recommend this treatment, at great cost to their income and careers?
Low-Quality Medical Guidelines Legitimize Potential Harm
When guideline recommendations are made without evidence, they are regarded as clinical consensus statements. In light of the lack of knowledge of safety or efficacy of treatment, one might wonder how there is clinical consensus on transgender treatment—that risky and irreversible treatments are the only treatment option to be researched, rather than considering whether thinking can be changed.
In fact, the WPATH SOC does allude to the possibility that therapy can be beneficial:
Often with the help of psychotherapy, some individuals integrate their trans- or cross-gender feelings into the gender role they were assigned at birth and do not feel the need to feminize or masculinize their body.
However, elsewhere they state that psychotherapy should focus on the patient’s distress, and not conversion therapy. There is no further discussion about researching how psychotherapy could help people avoid treatment altogether, despite the statement that psychotherapy helps some people integrate into their “gender role assigned at birth”.
The following quote in the Institute of Medicine document about poorly developed clinical guidelines holds true for the WPATH SOC regarding faulty guideline development:
Poorly articulated or indirect evidence chains can make it difficult to discern which parts of the analytic logic are based on science or opinion, the quality of that evidence, and how it was interpreted. Readers can be misled into thinking that there is more (or less) scientific support for recommendations than actually exists. The ambiguity can also cause difficulty in establishing research priorities.
Institute of Medicine
Many gender dysphoric youth and their parents wonder if therapy would be an alternative to permanent alterations to their bodies. They are told that therapy has been shown to be ineffective and that transition is the only cure for gender dysphoria. They are not being told the truth—that non-medical methods to deal with gender dysphoria have never been seriously researched.
Carl Heneghan, editor-in-chief of the British Medical Journal, and Professor of Evidence-Based Medicine from the University of Oxford performed a review of the literature for puberty blockers and cross-sex hormones in children and adolescence. He concluded: “The current evidence base does not support informed decision making and safe practice in children.”
The current evidence base does not support informed decision making and safe practice in children.
Carl Heneghan, editor-in-chief of the British Medical Journal, and Professor of Evidence-Based Medicine at the University of Oxford
The public (including media and other medical professional bodies) blindly trusts that the recommendations for transgender care have come out of robust scientific research and objective guideline development. However, even WPATH SOC committee members and transgender clinicians have repeatedly admitted that there is no long-term research to show that benefits outweigh harms in transgender treatment, whether it is social transitioning, puberty blockers, hormones or surgery.
In an article written by transgender clinicians and researchers five years AFTER the WPATH SOC was published (several of whom were on the WPATH SOC Committee) they state the lack of quality data in transgender research:
Expansion of the evidence-base to inform transgender clinical care requires rich systematically collected data that at present are scarce or lacking in the U.S. The available data pertaining to transgender health are often based on convenience samples and the majority of published studies in the U.S. are cross-sectional or retrospective. The few published prospective follow up studies are small and have examined a limited number of outcomes.
Three of the same gender professionals again said a year earlier in the AMA Journal of Ethics: “Transgender medicine presents a particular challenge for the development of evidence-based guidelines. First and foremost, data on health outcomes in transgender medicine are currently limited to retrospective studies, case series, and individual case reports.”
The same authors go on to say that, even though there is no evidence, “The existence of guidelines in the field of transgender medicine both legitimizes the need for gender-affirming medical and surgical interventions and informs medical practitioners and policymakers on how to best meet these needs.” This statement could not show more clearly the harm low-quality medical guidelines can do—legitimize potential harm and lead other medical professionals to assume the guidelines were developed based on quality data and scientific rigor.
Conclusion
When someone says “transition-related care is safe, effective, and supported by the entire mainstream of the medical community”, they are basing their faith unquestioningly on guidelines that were developed by people and organizations with conflicts of interest, with no systematic review, and with no evidence of safety or efficacy of treatment. These “guidelines” do not meet inclusion criteria for any clinical guideline database and have not received an endorsement from any professional body in Canada. And yet, WPATH guidelines are given as the rationale to support the unthinkable: to physically harm a distressed and vulnerable population.
To their credit, WPATH does not say anywhere in the SOC that treatment is proven safe and effective. Instead, the overall message in the guidelines is to urge physicians to follow the patient’s wishes only, putting aside scientific evidence and ethical delivery of care.
The concept of an innate gender identity is unproven, unverifiable, and does not even have a clear definition. In the same spirit of historical psychiatric misadventures, somatic treatments are being prescribed for a psychologically based identity disorder. How many vulnerable children, adolescents and young adults will be harmed before we realize that history is repeating itself.




A brilliant, much-needed analysis. This is crucial information that needs to be shared widely. Thank you.
I hope this has an impact. What could be done. IT feels like we are almost too late as their are so many deeply invested how do you step back in this situation?
Great question. It’s never too late. If anyone has ideas, we’re all ears! Make sure you subscribe to stay informed. We’ll be sharing more research and coming up with practical steps you can help us with.
Great article. I appreciate the research you guys are doing.
On a technical note: I had a super hard time reading it due to 8 lines being unreadable due to twitter/Facebook/Pinterest and other tabs blocking 3-4 letters on each line. I was forced to read the whole article in only 3-4 lines at the bottom of my screen that were unblocked. I had the same issues on the other genderreport.ca articles. Could these buttons be removed? It would make reading the articles much easier. Thanks
Thanks for the feedback! We’ll address that issue.
I would like to know if there are are any legal consequences for the doctors or or medical practitionersf if they they prescribe off label drugs 2 patients
Surely there is is a duty of care when prescribing drugs with unknown side effects and damaging side effects,
Doctors are using “informed consent” to waive their responsibility. The first legal challenge is happening in the UK by a detransitioner and former nurse at the gender identity clinic there. They’re arguing that people under the age of 18 are not capable of informed consent on this topic. In Canada, the BC Infants Act was used to uphold the right of a 14 year old to start cross-sex hormones against her father’s wishes. The court also ruled that the medical practitioners were acting competently because they were following WPATH. WPATH claims to be the “medical consensus” but my prediction is this will no longer be the case once there is a group of medical experts representing evidence-based approaches to care and taking a more careful look at the research. This is what is needed for lawsuits to be successful. Please subscribe to our mailing list to find out about upcoming developments in this area.
Absolutely but surely if they are using it waive responsibility then they are not truly informing the patient and therefore have not received true consent?
I know this is likely to be a legal issue or one for a medical ethics board but it stands to reason that consent cannot be given if they are not informed and we have seen time and time again that these patients are simply not being informed and in some cases flat out lied to. When the wave of legal battles coming are at an end I think we’ll see an absolute ban on this type of treatment. Here’s to hoping it’s sooner rather than later
An exceedingly helpful analysis of WPATH! This should be distributed far and wide, within parent groups and to politicians.
Great analysis, thank you. See this tweet for an update on WPATH SOC 7: “while promoting the upcoming version SOC8 due out soon, WPATH inadvertently admits the current SOC7 is not evidence-based (and apparently not standards of care at all)”; WPATH stated: “This version of the Standards of Care [8] is the first to be developed using an evidence-based approach.”
https://twitter.com/ZaneEmma/status/1315877083633995776
Thank-you for the enlightening article. Well laid out and considered. I’m sorry things have gotten to this point.
Excellent article; lots of examples and detail. Clearly written. Gives some hope that alternative voices are being published instead of censored.
Oh! But this was written in 2019. Things are much worse now?
Thank you “Pamela”.
I don’t think that it would ever be possible for an evidence driven team of scientists to create the guidlines to be followed. Researchers will be labeled as transphobic and will lose their reputation and status in short and long term by setting under scrutiny the current status quo. The only possibility that they will be respected is if their findings prove (almost) entirely the claims of WPATH which is very unlikely.
You raise a very interesting point. So if no “acceptable” evidence-based guidelines are possible the question comes down to an issue of consent. Just as the UK has ruled on the Keira Bell case, we’ll have to wait for a similar test here.
how would i do this? the doctors never check or cared. they never check my mental health history as it showed numerous red flags i went home from dr brassards clinic. ended up in the hospital 2 days later. everyone was there CAMH, PRIDE and the Oshawa mental health unit was involved. they all didnt know how this could have happened. 4 years later. I’m talking to camh now that states “were just a consulting hospital”. how do i fight this?
There is no profit in the evidence aka systemic review. This has been an issue throughout history and evolution of gender identity.
So, the very same conflict/bias that you have referred to in your article exists on the your side as well. To put it bluntly your the pot calling the kettle black.
With every medical procedure there are people who either don’t respond or respond adversely to treatment. Granted they are rare in all medical sectors including this one.
You can’t bubble wrap the world. I find it somewhat humorous that the detransitioners who fully transitioned are so vocal and idolized. Yes, they made a bad decision based on the facts they had at the time. But, going into it they at multiple points would have been questioned on their gamble. They were questioned at multiple points in their transition about whether or not they were sure about what they were doing regardless of counseling. Informed consent only applies to hormones.
It however doesn’t apply to surgeries unless you travel to a backwaters country with cash. If someone is that driven that after all the confirmation questions or paying cash continues on and ends up unhappy enough to do the same in reverse. Then its OK to feel bad for them, but, you should also be concerned for their mental health in the first place. That has Obsessive Compulsive Disorder or something similar written all over it.
As far as children and informed consent. I think that model should be discontinued for everything except for blockers without parental consent and 2 psychotherapist letter approvals. And blockers only with a documented history with a trained psychotherapist with a confirmation from a second. A kind of nuclear protocol because it has the same devastating effect long term if wrong.
I’m genuinely impressed with the clarity and depth of your writing. You’ve covered the topic comprehensively. Well done!